Australian pharmaceutical company Argent BioPharma has delivered its largest single commercial shipment of 1,000 units of CannEpil to Ireland, where the cannabinoid formulation for refractory epilepsy is fully covered under the National Health Insurance scheme, according to a The Australian report.
The shipment carries an approximate reimbursed retail value of A$783,000 (€468,000) and will be distributed by Irish commercial partner Georgelle Pharma in collaboration with European wholesaler and logistics partner Versa Pharmaceuticals. CannEpil carries a National Health Insurance retail reimbursement price of €467 per unit.
The delivery follows production of 1,000 units manufactured for the first time via Argent's partner under strict European Union standards, incorporating defined cannabinoid ratios with pharmaceutical-grade quality controls to ensure batch consistency, safety and dosing precision.
CannEpil is a pharmaceutical-grade, standardised cannabinoid formulation developed for managing seizures associated with refractory epilepsy, where seizures persist despite treatment with conventional anti-epileptic medicines. Approximately 30 per cent of patients fail to achieve adequate seizure control on standard therapies.
"The successful delivery further strengthened Argent BioPharma's European distribution platform, broadening reimbursed patient access and supporting CannEpil's continued commercialisation as a core neurology asset," said chief executive officer Roby Zomer. "Importantly, this delivery aligns operational execution, market access, and clinical positioning as we advance the next phase of the company's growth strategy."
The shipment demonstrates Argent's capability to execute scaled commercial supply under regulated pharmaceutical manufacturing pathways, supporting Ireland's position as a key European market for neurological therapies.
Explore the full detals of Argent BioPharma's CannEpil commercial expansion in Ireland in the complete report.



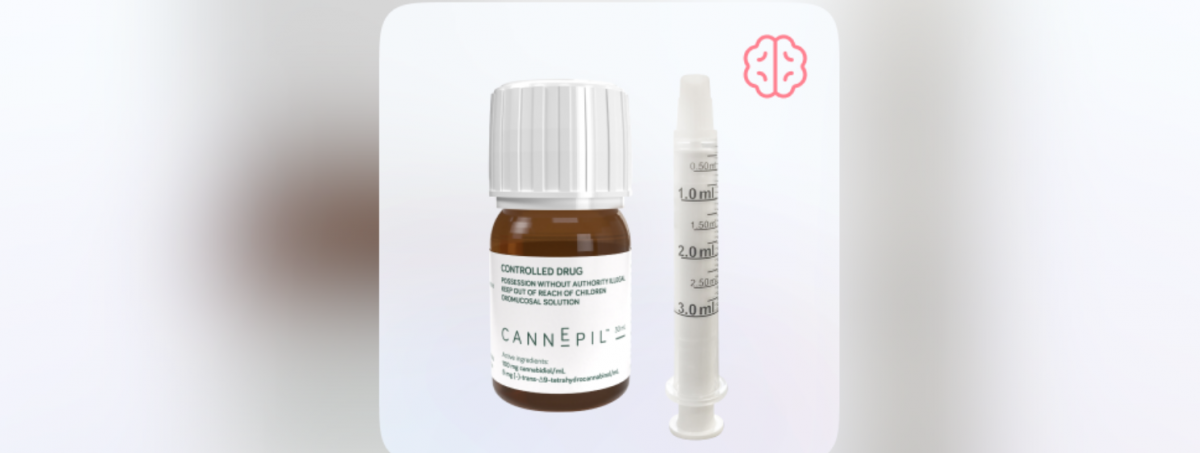















.png)

